
The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal
Introduction
In the year 2010, the incidence of newly diagnosed pancreatic cancer in USA was 43,140 and deaths attributable to pancreatic cancer were 18,770 (1). Among recently diagnosed pancreatic cancer patients, 65-70% will have advanced disease (stage III-IV) at initial presentation. Advanced pancreatic cancer has a very poor prognosis, with a median survival of 2-6 months for stage IV disease and 6-11 months for stage III disease. Overall, the 5-year survival among these patients is only 5-7% and the majority of patients survive less than 1-2 years. Even among patients who undergo surgery with curative-intent, >90% develops disease progression within 12-18 months. This poor prognosis is attributable to late stage presentation, lack of effective treatments, early recurrence and absence of clinically useful biomarker(s) which can detect pancreatic cancer in its precursor form(s) or earliest stages (2-4).
A wide-variety of tumor markers derived from serum, pancreatic tissue, pancreatic juice, saliva and/or stool has been proposed for early diagnosis as well as to predict prognosis in pancreatic cancer patients. Nevertheless, utility of those markers is often significantly limited by poor sensitivity, high false positive rates and lack of large scale validation (5).
Despite the vast number of potential pancreatic cancer biomarkers, very few have been thoroughly evaluated and none to the extent of carbohydrate antigen 19-9 (CA 19-9). This review provides a comprehensive review on the utility of serum CA 19-9 as a pancreatic cancer biomarker and its value in screening, diagnosis, staging, determination of resectability, early identification of recurrence and predicting treatment response.
Methods
A comphrensive literature search was performed using PubMed with keywords "pancreatic cancer" "tumor markers" "CA 19-9" "diagnosis" "screening" "prognosis" "resectability" and "recurrence". All English language articles pertaining to the role of CA 19-9 in pancreatic cancer for the years 1979-2010 were critically analyzed to determine its utility as a biomarker for pancreatic cancer.
Discussion
Koprowoski et al. first described CA 19-9 in colorectal cancer cell line (SW1116) using a monoclonal antibody (1116-NS-19-9) i.e. hybridoma technology in 1979 (6). CA 19-9 is also identified in the tissue and sera of patients with other gastrointestinal tumors including esophageal, gastric, biliary and pancreatic cancer (7). CA 19-9 also termed as sialyl Lewis-a (sLea), is expressed on the surface of cancer cells as a glycolipid and as an O-linked glycoprotein. CA 19-9 is derived from an aberrant pathway during production of its normal counterpart disialyl Lewis-a that has one extra sialic acid residue attached through a 2→6 linkage. Normally, Disialyl Lewis-a is expressed on the epithelial surface of digestive organs, acts as a ligand for monocytes and macrophages and helps in immunosurveillance. Epigenetic silencing of the gene for 2→6 sialyl transferase during early stages of carcinogenesis leads to abnormal synthesis and accumulation of sialyl Lewis-a (CA 19-9). sLea may also play a role in cancer invasion/metastasis as it is known to be a ligand for endothelial cell E-selectin responsible for cell adhesion (7-11).
CA 19-9 is related to the Lewis blood group antigens and only patients belonging to the Le (α-β+) or Le (α+β-) blood groups will express the CA 19-9 antigen (7). Le (α-β-) phenotypes occur in 5-10% of population which lack the enzyme 1,4-fucosyl transferase required for antigen epitope production, and as such limits the use of CA 19-9 as a universally applicable biomarker (12-15).
Utility of CA 19-9 serum levels as a diagnostic and screening marker for pancreatic cancer
An "ideal" tumor marker possesses high sensitivity enabling it to identify the disease in a screening population without symptoms. Several studies have explored the utility of CA 19-9 serum levels as a screening tool for pancreatic cancer in asymptomatic individuals as well as in patients with symptoms suspicious for pancreatic cancer (Table 1) (16,18,19). Kim et al. assessed CA 19-9 serum levels in 70,940 asymptomatic individuals and identified only 4 patients with pancreatic cancer among 1,063 patients with elevated CA 19-9 serum levels (>37 U/mL, mean values 50.5±16.8 U/mL) (17), yielding a dismal positive predictive value (PPV) of only 0.9%, although the sensitivity and specificity were 100 and 98.5% respectively. Satake et al. analyzed CA 19-9 serum levels in 12,840 asymptomatic and 8,706 individuals with symptoms suspicious for pancreatic cancer such as weight loss, epigastric pain and jaundice. These authors identified only 4 pancreatic cancers (1 resectable) among 18 asymptomatic patients (0.2%) with an elevated CA 19-9 serum level. Among the 8,706 patients with symptoms suspicious for pancreatic cancer, 198 patients (4.3%) had elevated CA 19-9 serum levels. Following extensive work up, 85 patients (1.8%) were found to have pancreatic cancer of which 28 patients (0.4%) were resectable (17). Similarly, Chang et al. have screened 5343 asymptomatic individuals for pancreatic cancer, and identified CA 19-9 serum level elevation (>37 U/mL) in 385 patients (7.2%) (18). Among this group only 2 patients (0.004%) had pancreatic cancer and their serum CA 19-9 levels were 88.4 U/mL and 46,885 U/mL respectively. The PPV of an elevated serum CA 19-9 level in the asymptomatic population in this study was only 0.5%. False positive elevation of the CA 19-9 serum levels was noted in 325 patients (6.1%) and a total of 58 other cancers were identified (16).
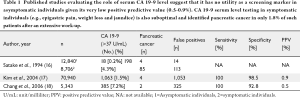
Full table
As evident from aforementioned studies, given the suboptimal sensitivity and poor predictive value of CA 19-9 serum levels and low prevalence of pancreatic cancer in the general population, routine serum CA 19-9 level testing has no utility as a screening tool in asymptomatic patients. Even among patients with symptoms suspicious for pancreatic cancer, elevated CA 19-9 serum levels is a poor predictor of pancreatic cancer with a predictive value of 0.5-0.9%. Equally noted in all of the screening studies is that a significant number of individuals with elevated CA 19-9 serum levels have actually harbored non-pancreatic neoplastic pathology which further undermines the applicability of serum CA 19-9 levels as a screening tool.
Among patients who present with a pancreatic mass, elevated CA 19-9 serum levels yield a much higher predictive value for diagnosing pancreatic cancer. Tessler et al. studied 150 patients undergoing surgery for suspected pancreatic cancer without a preoperative tissue diagnosis. Multivariate analysis identified that a combination of weight loss >20 lbs, bilirubin >3 mg/dL, and CA 19-9 >37 U/mL provided an almost 100% specificity and positive predictive value for pancreatic cancer regardless of the extent of imaging abnormalities (19).
Two previous reviews have attempted to summarize the diagnostic utility of CA 19-9 serum levels in patients with pancreatic cancer (14,20). Steinberg analyzed diagnostic value of CA 19-9 serum levels (37-40 U/mL) in 1040 patients (24 case series) with symptomatic pancreatic cancer and reported a median sensitivity and specificity of 81% and 90% respectively. The positive predictive value (PPV) and negative predictive value (NPV) of an elevated serum CA 19-9 level was 72.3% and 95.8% respectively. If the serum CA 19-9 threshold used to diagnose pancreatic cancer was raised to 100 U/mL or 1000 U/mL, the specificity increased to 98% and 99.8%, however the sensitivity decreased to 68% and 41% respectively (20). More recently, Goonetilleke et al. analyzed the utility of CA 19-9 serum levels (37-40 U/mL) to diagnose pancreatic cancer among 2,283 symptomatic patients reported in 26 case-series (17). In this report, the sensitivity and specificity of an elevated serum CA 19-9 level was 79% and 82% with a PPV and NPV of 72% and 81% respectively. Overall, an elevated serum CA 19-9 level has a sensitivity of 79-81% and a specificity of 82-90% for diagnosing pancreatic cancer in symptomatic patients (14).
Utility of CA 19-9 serum levels in assessment of pancreatic cancer stage and determination of surgical resectability
The value of pre-operative serum CA 19-9 levels to predict pancreatic cancer stage and determine resectability has been extensively studied (21-26) (Table 2). Kim et al. evaluated CA 19-9 serum levels in 114 pancreatic cancer patients who underwent either pancreatic resection (n=72) or palliative bypass surgery (n=42). These authors reported a positive correlation between pancreatic cancer stage and mean pre-operative CA 19-9 serum levels. In this study stage IA patients had a mean serum CA 19-9 level of 40.05 U/mL, stage IIA patients had mean serum levels of 469.64 U/mL, stage IIB patients had mean serum levels of 747.79 U/mL, stage III patients had mean serum levels of 709 U/mL, while stage IV patients had a mean serum CA 19-9 levels of 3239 U/mL (25). Safi et al. compiled preoperative CA 19-9 serum levels in 126 patients with resectable pancreatic cancer (22). In this study, 29 of 45 patients (64%) with stage I pancreatic cancer had elevated CA 19-9 with a median level of 68 U/mL (range, 9.0-3018 U/mL). Eight of 10 patients (80%) with stage II pancreatic cancer had elevated serum CA 19-9 level with a median levels of 72 U/mL (range, 8.4-5000 U/mL). Eighty one percent (47 out of 58) of patients with stage III disease had an elevated CA 19-9 levels (median, 210 U/mL, range, 2-7496 U/mL) and 100% of patients (n=13) with stage IV disease had an elevated CA 19-9 serum levels (median 412 U/mL, range, 49.6-14,600 U/mL). In an effort to correlate advanced stage disease with higher CA 19-9 serum levels, these authors also noted that an elevated pretreatment CA 19-9 serum level of ≥300 U/mL indicated unresectable disease in 80% of patients. However the above correlation between CA 19-9 serum levels and pancreatic cancer resectability is not universal but undermined by the fact that 5-10% of patients with pancreatic cancer will not demonstrate elevated serum CA 19-9 serum levels given their sialyl Lewis negative state and by false positive elevations in obstructive jaundice (7). Moreover, CA 19-9 serum levels alone should not be the sole criteria used in making decisions to proceed to surgery; rather CA 19-9 serum levels is one of several contributing factors used in combination with clinical evaluation and information obtained from radiological and endoscopic imaging.

Full table
Anatomic imaging provides vital information regarding local invasiveness of pancreatic cancer and the presence of metastatic disease. Recent advances in radiologic (CT scan), Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET scan) and endoscopic imaging [Endoscopic Ultrasound (EUS), Endoscopic Retrograde Cholangiopancreatography (ERCP)] and increased use of staging laparoscopy have enabled better delineation and staging of pancreatic cancer, which in turn has helped to reduce the negative laparotomy rate (27,28). Despite those advancements, up to 15% of patients with pancreatic cancer are found unresectable at the time of surgery, which is attributable to occult vascular invasion, presence of undetected metastasis or positive peritoneal lavage cytology (25). Whether pre-operative CA 19-9 serum levels can be used as a surrogate marker for tumor resectability has been extensively evaluated (21,27-29) (Table 3). Schleiman et al. evaluated preoperative CA 19-9 serum levels in 89 pancreatic cancer patients prior to surgical exploration and noted that mean CA 19-9 serum levels were significantly lower in resectable tumors compared to those with locally advanced tumors (63 vs. 592 U/mL, P=0.003) or with metastatic disease (63 vs. 1387 U/mL, P<0.001) (32) (Table 3). A pre-operative CA19-9 serum level of >150 U/mL was associated with an 88% positive predictive value for unresectability, whereas serum levels <150 U/mL had a negative predictive value of 64% (32). Kim et al. evaluated CA 19-9 serum levels in 72 patients treated surgically for "resectable" pancreatic adenocarcinoma and 42 patients treated with surgical palliation (bypass surgery). The median CA 19-9 serum levels for patients achieving an R0 resection, R1 resection or R2 resection, was 49.66, 233.0 and 600 U/mL respectively. The median CA 19-9 serum level for patients with peritoneal metastasis was 780.49 U/mL. These authors concluded that a pre-operative CA 19-9 ≥92.77 U/mL predicted an R1/2 resection or unresectability with a 90.6% accuracy. It is important to note however that lower pre-operative CA 19-9 serum levels predicted the probability of an R0 resection in only 27.1% of patients (25). In summary, these studies suggest that a median CA 19-9 serum level <100 U/mL correlates with resectability (41-80%) whereas levels >100 U/mL suggest advanced or metastatic pancreatic cancer (60-85%) (22,25,29-37)(Table 3). Nevertheless, 10-15% of patients with a low or normal pre-operative CA 19-9 serum levels may harbor unresectable disease identified at exploration, similarly 5-10% of patients with elevated pre-operative CA 19-9 serum level will be resectable (12,15). Halloran et al. identified unresectable disease in 17 out of 80 (21%) patients with low CA 19-9 serum levels (<37 U/mL) who were deemed resectable by radiologic criteria (37). While the pre-operative serum CA 19-9 level provides a good prognostic information on pancreatic cancer stage, however, it should not be the sole criteria for determining resectability to avoid false negative or false positive surgical exploration (15,27,28).
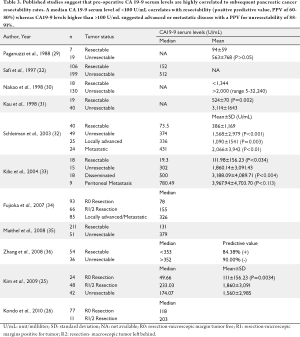
Full table
Utility of CA 19-9 serum levels as a biomarker of prognosis in patients with pancreatic cancer
The value of serum CA 19-9 levels to provide meaningful prognostic information and permit patient stratification (survival groups) based on its serum level has been extensively investigated (22,24,26,30,31,38-49)(Table 4). Waraya et al. performed a multivariate analysis of factors predicting survival in 117 pancreatic cancer patients undergoing surgical resection and reported that a low preoperative CA 19-9 serum levels (28-30 U/mL) [P=0.006, relative risk (RR), 2.16] and positive peripancreatic margin (P=0.04, RR, 1.62) independently predicted survival (46). Moreover they noted that the higher the preoperative CA19-9 serum level, the worse the prognosis. Patients with a preoperative CA 19-9 serum levels of <37 U/mL (n=23) had a 5-year disease specific survival (DSS) of 60.0% compared to 4.0% DSS among patients with CA 19-9 serum levels >37 U/mL (n=66) (P=0.0001). Even more notable was the fact that 76.9% of stage III pancreatic cancer patients with a CA19-9 serum level of <37 U/mL survived more than 5 years (average DSS of 26.9 months). Barugola et al. analyzed factors predictive of early death (within 12 months) among 224 surgically resected pancreatic cancer patients and reported that an elevated preoperative CA 19-9 serum levels of >200 U/mL, a high grade tumor, an R2 resection and prolonged symptoms independently predicted early death (within 12 months) (46). Berger et al. stratified 129 surgically resected pancreatic cancer patients into 4 groups based on their pre-operative CA 19-9 level [(undetectable, normal (<37 U/mL), 38-200 U/mL, and >200 U/mL)]. Patients with undetectable pre-operative CA 19-9 serum levels and those with levels of <37 U/mL had an improved median survival (32 and 35 months, respectively) compared to patients with CA 19-9 serum levels between 38-200 U/mL or >200 U/mL (22 and 16 months, respectively) (43). Smith et al. evaluated preoperative CA 19-9 serum levels in 109 pancreatic cancer patients who underwent a pancreatoduodenectomy and noted a median survival of only 10.4 months in patients with a preoperative CA19-9 level >150 U/mL (n=64), compared to a median survival of 22.1 months in patients with a CA19-9 serum level ≤150 U/mL (n=45, P=0.012) (45). Table 3 lists additional studies which have used various cut-off levels for pre-operative CA 19-9 serum levels in an effort to predict survival among pancreatic cancer patients (22,24,26,30,31,38-49). These studies support the conclusion that a normal (<37 U/mL) or low preoperative CA 19-9 serum level (<100 U/mL) correlates with early pancreatic cancer stage and independently predicts improved overall survival, whereas an elevated CA 19-9 serum levels (>100 U/mL) is associated with a poor prognosis (38-49).
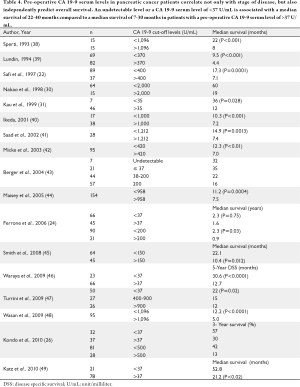
Full table
Several authors have reported on the prognostic significance of the post-operative CA 19-9 serum levels in predicting survival. Ferrone et al. analyzed 111 pancreatic cancer patients in whom pre- and post-operative CA 19-9 serum levels were measured. Post-operative CA 19-9 serum levels of <37 U/mL were associated with a mean survival of 2.4 years, a level of <200 U/mL had a mean survival of 2.3 years, whereas a post-operative CA 19-9 serum levels of <1000 U/mL and >2000 U/mL had a mean survival of 9 and 5 months respectively. Overall a low postoperative serum CA 19-9 level (<200 U/mL) was an independent predictor of survival (24).
Kondo et al. studied pre- and postoperative CA19-9 serum levels in 109 surgically treated pancreatic cancer patients and identified that both a normal postoperative CA 19-9 serum level (37 U/mL) [Hazard Ratio (HR) 1.64, P=0.004], and the addition of adjuvant chemotherapy were independent predictors of prognosis (26). More specifically these authors identified that a post-operative CA 19-9 serum level measured at 2-5 weeks could independently predict a prolonged 3- year survival rate. Post-operative CA 19-9 serum levels of <37 U/mL, <200 U/mL and >500 U/mL were associated with a 49%, 38%, and 0% 3-year survival rates respectively. Elevated CA 19-9 (>35 U/mL) in the immediate post-operative period was also associated with an R1 resection and lymph node metastases (P=0.041) (26). Montgomery et al. assessed 40 pancreatic cancer patients who had undergone surgical resection and found that patients in whom the CA 19-9 serum levels returned to normal within the first postoperative year had a longer overall survival compared to patients in whom CA 19-9 serum levels remained elevated (34 vs.13 months, P<0.04) (50-52). Given the half life of CA 19-9 is approximately 14 hours, those authors suggested that post-operative CA 19-9 serum levels should be measured 4-6 weeks following surgery and that patients with elevated levels are likely to harbor residual tumor or sub-clinical metastases. In summary, postoperative normalization or a downward trend of the CA 19-9 serum level following pancreatic resection is associated with prolonged survival whereas elevated or failure of the CA 19-9 to decrease following pancreatic resection reflects residual disease or occult metastasis and portends a poor survival.
Utility of CA 19-9 serum levels to assess response to chemotherapy in pancreatic cancer patients
Most patients with pancreatic cancer require chemotherapy and/or radiation, either in the neo-adjuvant setting to improve resectability or treat suspected micro-metastasis, or in the adjuvant setting for locally advanced disease, high grade tumor and when vascular invasion or lymph node metastases are present. Whether serum CA 19-9 levels can be used as a surrogate marker of response to chemotherapy has been studied in a variety of clinical settings (41,44,53-64). Willett et al. measured CA 19-9 serum levels in 42 resectable pancreatic cancer patients receiving neoadjuvant treatment with 5-flourouracil and external beam radiation prior to planned pancreaticoduodenectomy. Among 10 patients with an increased CA 19-9 serum level following treatment, 9 (90%) had distant metastases or local tumor progression. In contrast, only 6 of 29 patients (21%) with a declining CA 19-9 serum level after neo-adjuvant chemo-radiotherapy had metastases or local tumor progression on restaging CT scan or at laparotomy. Whether the CA 19-9 serum level increased or decreased during treatment, correlated significantly with disease progression (P=0.009) (65). Katz et al. studied 119 patients with pancreatic cancer who were treated with neoadjuvant chemotherapy followed by pancreaticoduodenectomy. These authors found that a post-treatment CA 19-9 serum level of <37 U/mL had an 86% PPV for successful completion of the pancreatic resection, and a NPV of only 33%. Post-treatment CA 19-9 serum levels <61 U/mL also had a high 93% PPV but a diminishing 28% NPV in regards to predicting successful completion of pancreaticoduodenectomy among resectable patients (49). Although post-treatment CA 19-9 serum levels in the above mentioned study had a high PPV in regards to likelihood of resectability following neo-adjuvant chemotherapy, the low NPV highlights the importance of re-staging radiographic evaluation as well as laparoscopy prior to surgical exploration (34,49).
Several authors have reported on the use of CA 19-9 serum level trends to assess chemotherapy response using such definitions as ≥20% or ≥50-75% decline in CA 19-9 serum levels within the first 6-8 weeks of treatment. Nearly all studies have demonstrated that a treatment related decline in CA 19-9 serum levels is associated with prolonged survival and is an independent predictor of overall survival (41,44,53-64) (Table 5). Reni et al. compared basal CA 19-9 serum levels in 247 advanced pancreatic cancer patients enrolled in 5 consecutive chemotherapy trials (G, gemcitabine; PEFG, cisplatin, epirubicin, 5-fluorouracil, and gemcitabine; PDXG, cisplatin, docetaxel, capecitabine, and gemcitabine) (60). The survival curves were plotted based on a pre-defined decline in CA 19-9 serum levels (Group 1, <50% decrease, Group 2, 50% to 89% decrease and Group 3, >89% decrease). Patients with a higher percent decline in CA 19-9 serum level following treatment had improved overall survival (Group III-16.7 months compared to Group II-10 months, P=0.002, and Group II- 10 months vs. 6.5 months for Group -I, P=0.002). Overall, the median survival was 15.5 months among patients with normal CA 19-9 levels, 11.9 months among 108 patients with CA 19-9 serum levels between 38 U/mL and 1167 U/mL and 8 months among 105 patients who had CA 19-9 serum levels >1167 U/mL (60).
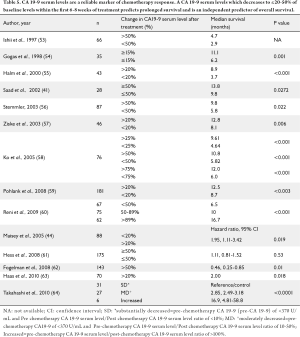
Full table
Halm et al. evaluated CA 19-9 serum levels in 36 patients enrolled in gemcitabine chemotherapy trials and reported that patients with a decline in CA 19-9 serum levels of >20% from baseline after 8 weeks of treatment (n=25) had improved median survival compared to patients with a rise or a decrease of <20% (n=11) (268 vs. 110 days, P=0.001) (55). Moreover, treatment related decline in CA 19-9 serum levels was the strongest independent predictor of survival (P<0.001) on multivariate analysis. Finally, using a novel approach to compute log CA 19-9 kinetics among 115 patients enrolled in first line pancreatic cancer chemotherapy, Boeck et al. demonstrated that log CA 19-9 kinetics was a significant predictor of both time to tumor progression (Hazard Ratio, HR 1.48, P<0.001) and overall survival (HR 1.34, P<0.001) (66).
Utility of CA 19-9 serum levels to predict post-operative recurrence
The predictive value of current methods (CT scan and PET scan) to assess early post-operative recurrence is sub-optimal given that pancreatic resection is often associated with intense desmoplastic and post-operative inflammatory changes leading to dense fibrosis making radiological detection difficult (15,41,60). The utility of sequential post-operative CA 19-9 serum level measurement to detect early recurrence in pancreatic cancer patients has been well studied. Kang et al. evaluated factors predictive of post-operative recurrence in 61 pancreatic cancer patients and reported that an adjusted CA 19-9 serum level (defined as a ratio of CA 19-9 serum levels divided by serum bilirubin when higher than 2 mg/dL) of >50 U/mL was associated with an increased recurrence risk (twice) when compared to adjusted levels of <50 U/mL (67). Montgomery et al. reported that a significant and sustained post-operative elevations of CA 19-9 serum levels preceded clinical or radiologic detection of recurrence by 2 weeks to 5 months (median 3.5 months) and that an elevated post-operative CA 19-9 serum levels >180 U/mL was associated with a disease free survival of 12 months compared to 35 months for patients with post-operative CA 19-9 serum levels <180 U/mL (50). In this study, patients whose postoperative CA 19-9 values normalized by 3 to 6 months (<37 U/mL) had a longer disease free survival (24 vs. 10 months, P<0.04) and median survival (34 vs. 13 months, P<0.04). Hernandez et al. analyzed data from 96 surgically resected pancreatic cancer patients in whom CA 19-9 serum levels were drawn at baseline, 4 weeks, and 12-week intervals following surgery and for whom CA 19-9 velocity was calculated (rate of change in CA 19-9 levels over a 4-week period). These authors found that CA 19-9 velocity was a better predictor of overall survival than baseline CA 19-9 serum levels (P<0.001). Patients with disease progression had a CA 19-9 velocity of 131 U/mL/4-weeks compared to a velocity of 1 U/ml/4-weeks at 22 months for patients without disease progression (P<0.001) (51). In summary, the above results imply that clinical or radiologic post-operative recurrence is often preceded or associated with elevated CA 19-9 serum levels by 2-6 months. Elevation of post-operative CA 19-9 serum levels or failure of the CA 19-9 serum levels to normalize in the post-operative period suggest the presence of residual tumor or remnant disease and is associated with a poor prognosis.
Limitations that undermine the utility of CA 19-9 serum level as a preferred tumor marker for pancreatic cancer.
Despite multiple clinical applications for CA 19-9 serum levels in pancreatic cancer patients, the diagnostic utility of CA 19-9 is limited due to a low or modest sensitivity (79-81%) in symptomatic patients and a low PPV (0.9%) which makes it suboptimal screening test (12,14,16,18,19). Even among individuals at higher risk of pancreatic cancer (hereditary pancreatitis, family history of pancreatic cancer, Peutz-Jeghers syndrome), CA 19-9 serum levels fail to identify early/small tumors or precancerous lesions in 10-15% of patients (68), is elevated in only 80-85% of pancreatic cancer patients (12,14,20). The CA 19-9 serum levels are not predictive of tumor location or differentiation. As noted earlier, CA 19-9 serum levels may be elevated in a variety of non-pancreatic neoplastic conditions resulting in a high false positive rate (10-30%). Benign conditions associated with elevated serum CA 19-9 levels include ovarian cyst, heart failure, hashimoto's thyroiditis, rheumatoid arthritis and diverticulitis (16-19,69-74) (Table 6). Marked elevations in CA 19-9 serum levels have also been reported in numerous benign and malignant biliary conditions (15-38.8%) such as choledocholithiasis, gallbladder cancer and cholangiocarcinoma. Finally, CA 19-9 serum levels alone cannot differentiate between benign, precursor lesions and malignant pancreatic conditions such as acute and chronic pancreatitis, intraductal pancreatic mucinous neoplasms (IPMN), pancreatic intra-epithelial neoplasia (PANIN) and pancreatic cancer, as the former are also associated with elevated CA 19-9 serum levels in 10-50% of cases (69-75).
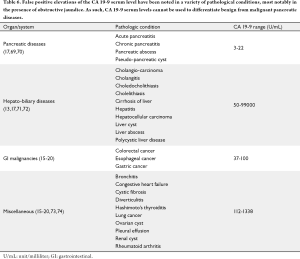
Full table
Hyperbilirubinemia is also a significant confounding factor since it is associated with an increased CA 19-9 serum level in cases of both benign and malignant biliary obstruction (8,9,12,20). Although CA 19-9 serum levels in the presence of obstructive jaundice may have higher sensitivity, it is at the cost of decreased specificity and accuracy. Mery et al. studied 548 patients with obstructive jaundice and reported a higher CA 19-9 serum level among pancreatic cancer patients compared to those with other hepatobiliary malignancies or benign diseases. These authors noted that by increasing the cut-off level for CA 19-9 serum level from 37 to 90 U/mL they were better able to differentiate malignant hepatobiliary diseases from benign diseases (sensitivity 86% vs. 61% and specificity 39% vs. 86%) (75). Kau et al. studied 86 resectable and 57 unresectable pancreatic cancer patients and reported that a mean CA 19-9 serum levels of 191±6 U/mL and 1203±400 U/mL was associated with serum bilirubin levels of <7.3 mg/dL or >7.3 mg/dl respectively (31). Ong et al. studied 83 patients presenting with abnormal CA19-9 serum levels and radiological or clinical features suggestive of hepato-biliary-pancreatic (HPB) malignancy who were subsequently found to have benign disease. On multivariate analysis, these authors reported that hyperbilirubinemia (serum bilirubin >2 mg/dL) was an independent factor predictive of CA 19-9 serum level (P=0.028) (76,77).
Biliary drainage which results in a decrease in CA 19-9 serum levels may suggest benign conditions. Marrelli et al. studied 128 patients admitted with obstructive jaundice including 87 patients with pancreatico-biliary malignancy and 42 patients with benign diseases. CA 19-9 serum levels were elevated in 61% of benign causes and 86% of malignant causes, which resulted in a reduction in accuracy to 61%. Following biliary drainage CA 19-9 serum levels decreased in nearly all benign cases (41 of 42 patients, 98%) but in only 19 out of 38 (50%) patients with malignant biliary obstruction (78). Kau et al. reported a 40% reduction in CA 19-9 serum levels after relief of malignant biliary obstruction. Several authors have postulated that inflammation associated with obstructive jaundice increases proliferation of biliary epithelial cells with a subsequent increase in systemic absorption of CA 19-9. The CA 19-9 serum levels normalize after treatment of benign cholestasis, whereas it remains elevated in malignant obstruction due to persistent production of CA 19-9 by proliferating tumor cells (31).
In an effort to increase the specificity and accuracy of CA 19-9 serum evaluation in the setting of hyperbilirubinemia, several authors have suggested using higher cut-off levels for serum CA 19-9 or choosing a level determined by receptor operator characteristic (ROC) curves associated with higher specificity. Marrelli et al. evaluated an increased serum CA 19-9 cut-off level of 90 U/mL, and noted that the specificity increased to 95%, while the sensitivity declined to 61% (78). Similarly, using a CA 19-9 serum cut-off level of >1,000 U/mL in the presence of hyperbilirubinemia, Kim et al. reported a specificity of nearly 100%, but a sensitivity of less than 50% (25). Ortiz-Gonzalez et al. studied 26 patients with resectable pancreatic cancer and found that the median adjusted CA 19-9 serum level was significantly lower (P=0.01) among patients with normal biliary excretion than those with bilirubin levels >2 mg/dL (79). Kang et al. assessed the value of adjusted CA 19-9 serum levels to predict post-operative recurrence in 61 patients who underwent pancreatic resection. Adjusted preoperative CA 19-9 serum levels were significantly lower compared to baseline CA 19-9 serum levels (129.4±225.2 U/mL vs. 442.1±645.5 U/mL, P<0.0001). In this study an adjusted preoperative CA 19-9 serum level of ≥50 U/mL (P=0.027) was an independent predictive factor for tumor recurrence (67). Contrary to the above findings, a recent article reported no effect of hyperbilirubinemia on CA 19-9 serum levels. Maithel et al. studied 491 patients in whom preoperative CA 19-9 serum level was evaluated to predict presence of sub-radiographic unresectable disease at the time of staging laparoscopy. These authors failed to find any significant correlation between CA 19-9 serum levels and elevated bilirubin levels (Pearson correlation coefficient 0.12) irrespective of tumor location (pancreatic head or body/tail) (35).
Despite the anomalous report cited above, CA 19-9 serum levels are often significantly elevated in the setting of obstructive jaundice, resulting in a further increase in false positives in benign conditions thereby reducing the overall accuracy and specificity of CA 19-9 as a diagnostic marker. The use of adjusted CA 19-9 serum levels or using higher CA 19-9 cut-off levels in the setting of hyper-bilirubinemia and re-evaluation of CA 19-9 serum levels following the treatment of obstruction should improve the diagnostic utility.
Finally, as mentioned earlier, sialyl Lewis negative phenotype seen in 5-10% of population is associated with false negative results for CA 19-9 serum levels even in the presence of advanced pancreatic cancer (7). Other biomarkers such as duke pancreatic monoclonal antigen type 2 (DUPAN-2), macrophage inhibitory cytokine (MIC-1), regenerating islet derived (REG-4) which are unaffected by Lewis blood group status may be more effective for this population (7,80,81). Additional strategies include simultaneous measurement of disialyl Lewis a (normal counterpart) during CA 19-9 evaluation. The ratio of sLea (CA 19-9)/disialyl Lewis may provide an improved serum diagnosis by averting undesired effect of a Lewis-blood group negative phenotype and reducing the false-positive rate (non-specific elevation) (7).
Conclusions
Pancreatic cancer is associated with a dismal prognosis and biomarkers that can detect pancreatic cancer in its earliest stages should improve prognosis. Despite a large number of putative biomarkers for pancreatic cancer, carbohydrate antigen (CA 19-9) is the most extensively studied and currently the gold-standard biomarker for pancreatic cancer diagnosis in symptomatic patients. Pre-operative CA 19-9 serum levels provide important prognostic information in pancreatic cancer patients, correlate with tumor stage and independently predict overall survival. An increasing postoperative CA 19-9 serum level or failure of the CA 19-9 serum levels to normalize post-operatively is associated with a poor prognosis and suggests residual disease or the presence of occult metastases, while a decline or normalization of the post-operative CA 19-9 serum level, is associated with improved survival. CA 19-9 serum levels assessment can be used as a surrogate marker of response to chemotherapy with a ≥ 20-50% decrease in CA 19-9 serum levels following chemotherapy associated with a positive tumor response and increased survival. Limitations such as false negative results in sialyl Lewis negative individuals and false positive elevation in the presence of obstructive jaundice limit the universal applicability of serum CA 19-9 and the poor PPV of CA 19-9 serum level renders it impotent as a screening tool.
Footnote
No potential conflict of interest.
References
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin 2010;60:277-300. [PubMed]
- Ellison LF, Wilkins K. An update on cancer survival. Health Rep 2010;21:55-60. [PubMed]
- Maisonneuve P, Lowenfels AB. Epidemiology of pancreatic cancer: an update. Dig Dis 2010;28:645-656. [PubMed]
- Li J, Merl MY, Chabot J, Saif MW. Updates of adjuvant therapy in pancreatic cancer: where are we and where are we going? Highlights from the "2010 ASCO Annual Meeting". Chicago, IL, USA. June 4-8, 2010. JOP 2010;11:310-312. [PubMed]
- Harsha HC, Kandasamy K, Ranganathan P, et al. A compendium of potential biomarkers of pancreatic cancer. PLoS Med 2009;6:e1000046. [PubMed]
- Koprowski H, Steplewski Z, Mitchell K, Herlyn M, Herlyn D, Fuhrer P. Colorectal carcinoma antigens detected by hybridoma antibodies. Somatic Cell Genet 1979;5:957-971. [PubMed]
- Kannagi R. Carbohydrate antigen sialyl Lewis a--its pathophysiological significance and induction mechanism in cancer progression. Chang Gung Med J 2007;30:189-209. [PubMed]
- Safi F, Roscher R, Bittner R, Schenkluhn B, Dopfer HP, Beger HG. High sensitivity and specificity of CA 19-9 for pancreatic carcinoma in comparison to chronic pancreatitis. Serological and immunohistochemical findings. Pancreas 1987;2:398-403. [PubMed]
- Duraker N, Hot S, Polat Y, Höbek A, Gençler N, Urhan N. CEA, CA 19-9, and CA 125 in the differential diagnosis of benign and malignant pancreatic diseases with or without jaundice. J Surg Oncol 2007;95:142-147. [PubMed]
- Liao Q, Zhao YP, Yang YC, Li LJ, Long X, Han SM. Combined detection of serum tumor markers for differential diagnosis of solid lesions located at the pancreatic head. Hepatobiliary Pancreat Dis Int 2007;6:641-645. [PubMed]
- Vestergaard EM, Hein HO, Meyer H, et al. Reference values and biological variation for tumor marker CA 19-9 in serum for different Lewis and secretor genotypes and evaluation of secretor and Lewis genotyping in a Caucasian population. Clin Chem 1999;45:54-61. [PubMed]
- Ritts RE, Pitt HA. CA 19-9 in pancreatic cancer. Surg Oncol Clin N Am 1998;7:93-101. [PubMed]
- Kim HJ, Kim MH, Myung SJ, et al. A new strategy for the application of CA19-9 in the differentiation of pancreaticobiliary cancer: analysis using a receiver operating characteristic curve. Am J Gastroenterol 1999;94:1941-1946. [PubMed]
- Goonetilleke KS, Siriwardena AK. Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur J Surg Oncol 2007;33:266-270. [PubMed]
- Duffy MJ, Sturgeon C, Lamerz R, et al. Tumor markers in pancreatic cancer: a European Group on Tumor Markers (EGTM) status report. Ann Oncol 2010;21:441-447. [PubMed]
- Kim JE, Lee KT, Lee JK, Paik SW, Rhee JC, Choi KW. Clinical usefulness of carbohydrate antigen 19-9 as a screening test for pancreatic cancer in an asymptomatic population. J Gastroenterol Hepatol 2004;19:182-186. [PubMed]
- Steinberg W. The clinical utility of the CA 19-9 tumor-associated antigen. Am J Gastroenterol 1990;85:350-355. [PubMed]
- Satake K, Takeuchi T, Homma T, Ozaki H. CA19-9 as a screening and diagnostic tool in symptomatic patients: the Japanese experience. Pancreas 1994;9:703-706. [PubMed]
- Chang CY, Huang SP, Chiu HM, Lee YC, Chen MF, Lin JT. Low efficacy of serum levels of CA 19-9 in prediction of malignant diseases in asymptomatic population in Taiwan. Hepatogastroenterology 2006;53:1-4. [PubMed]
- Tessler DA, Catanzaro A, Velanovich V, Havstad S, Goel S. Predictors of cancer in patients with suspected pancreatic malignancy without a tissue diagnosis. Am J Surg 2006;191:191-197. [PubMed]
- Pleskow DK, Berger HJ, Gyves J, Allen E, McLean A, Podolsky DK. Evaluation of a serologic marker, CA19-9, in the diagnosis of pancreatic cancer. Ann Intern Med 1989;110:704-709. [PubMed]
- Safi F, Schlosser W, Kolb G, Beger HG. Diagnostic value of CA 19-9 in patients with pancreatic cancer and nonspecific gastrointestinal symptoms. J Gastrointest Surg 1997;1:106-112. [PubMed]
- Jiang XT, Tao HQ, Zou SC. Detection of serum tumor markers in the diagnosis and treatment of patients with pancreatic cancer. Hepatobiliary Pancreat Dis Int 2004;3:464-468. [PubMed]
- Ferrone CR, Finkelstein DM, Thayer SP, Muzikansky A, Fernandez-delCastillo C, Warshaw AL. Perioperative CA19-9 levels can predict stage and survival in patients with resectable pancreatic adenocarcinoma. J Clin Oncol 2006;24:2897-2902. [PubMed]
- Kim YC, Kim HJ, Park JH, et al. Can preoperative CA19-9 and CEA levels predict the resectability of patients with pancreatic adenocarcinoma? J Gastroenterol Hepatol 2009;24:1869-1875. [PubMed]
- Kondo N, Murakami Y, Uemura K, et al. Prognostic impact of perioperative serum CA 19-9 levels in patients with resectable pancreatic cancer. Ann Surg Oncol 2010;17:2321-2329. [PubMed]
- DeWitt J, Devereaux B, Chriswell M, et al. Comparison of endoscopic ultrasonography and multidetector computed tomography for detecting and staging pancreatic cancer. Ann Intern Med 2004;141:753-763. [PubMed]
- Ritts RE Jr, Nagorney DM, Jacobsen DJ, Talbot RW, Zurawski VR Jr. Comparison of preoperative serum CA19-9 levels with results of diagnostic imaging modalities in patients undergoing laparotomy for suspected pancreatic or gallbladder disease. Pancreas 1994;9:707-716. [PubMed]
- Paganuzzi M, Onetto M, Marroni P, et al. CA 19-9 and CA 50 in benign and malignant pancreatic and biliary diseases. Cancer 1988;61:2100-2108. [PubMed]
- Nakao A, Oshima K, Nomoto S, et al. Clinical usefulness of CA-19-9 in pancreatic carcinoma. Semin Surg Oncol 1998;15:15-22. [PubMed]
- Kau SY, Shyr YM, Su CH, Wu CW, Lui WY. Diagnostic and prognostic values of CA 19-9 and CEA in periampullary cancers. J Am Coll Surg 1999;188:415-420. [PubMed]
- Schlieman MG, Ho HS, Bold RJ. Utility of tumor markers in determining resectability of pancreatic cancer. Arch Surg 2003;138:951-955, discussion 955-956. [PubMed]
- Kiliç M, Göçmen E, Tez M, Ertan T, Keskek M, Koç M. Value of preoperative serum CA 19-9 levels in predicting resectability for pancreatic cancer. Can J Surg 2006;49:241-244. [PubMed]
- Fujioka S, Misawa T, Okamoto T, et al. Preoperative serum carcinoembryonic antigen and carbohydrate antigen 19-9 levels for the evaluation of curability and resectability in patients with pancreatic adenocarcinoma. J Hepatobiliary Pancreat Surg 2007;14:539-544. [PubMed]
- Maithel SK, Maloney S, Winston C, et al. Preoperative CA 19-9 and the yield of staging laparoscopy in patients with radiographically resectable pancreatic adenocarcinoma. Ann Surg Oncol 2008;15:3512-3520. [PubMed]
- Zhang S, Wang YM, Sun CD, Lu Y, Wu LQ. Clinical value of serum CA19-9 levels in evaluating resectability of pancreatic carcinoma. World J Gastroenterol 2008;14:3750-3753. [PubMed]
- Halloran CM, Ghaneh P, Connor S, Sutton R, Neoptolemos JP, Raraty MG. Carbohydrate antigen 19.9 accurately selects patients for laparoscopic assessment to determine resectability of pancreatic malignancy. Br J Surg 2008;95:453-459. [PubMed]
- Sperti C, Pasquali C, Catalini S, et al. CA 19-9 as a prognostic index after resection for pancreatic cancer. J Surg Oncol 1993;52:137-141. [PubMed]
- Lundin J, Roberts PJ, Kuusela P, Haglund C. The prognostic value of preoperative serum levels of CA 19-9 and CEA in patients with pancreatic cancer. Br J Cancer 1994;69:515-519. [PubMed]
- Ikeda M, Okada S, Tokuuye K, Ueno H, Okusaka T. Prognostic factors in patients with locally advanced pancreatic carcinoma receiving chemoradiotherapy. Cancer 2001;91:490-495. [PubMed]
- Saad ED, Machado MC, Wajsbrot D, et al. Pretreatment CA 19-9 level as a prognostic factor in patients with advanced pancreatic cancer treated with gemcitabine. Int J Gastrointest Cancer 2002;32:35-41. [PubMed]
- Micke O, Bruns F, Schäfer U, Kurowski R, Horst E, Willich N. CA 19-9 in the therapy monitoring and follow-up of locally advanced cancer of the exocrine pancreas treated with radiochemotherapy. Anticancer Res 2003;23:835-840. [PubMed]
- Berger AC, Meszoely IM, Ross EA, Watson JC, Hoffman JP. Undetectable preoperative levels of serum CA 19-9 correlate with improved survival for patients with resectable pancreatic adenocarcinoma. Ann Surg Oncol 2004;11:644-649. [PubMed]
- Maisey NR, Norman AR, Hill A, Massey A, Oates J, Cunningham D. CA19-9 as a prognostic factor in inoperable pancreatic cancer: the implication for clinical trials. Br J Cancer 2005;93:740-743. [PubMed]
- Smith RA, Bosonnet L, Ghaneh P, et al. Preoperative CA19-9 levels and lymph node ratio are independent predictors of survival in patients with resected pancreatic ductal adenocarcinoma. Dig Surg 2008;25:226-232. [PubMed]
- Waraya M, Yamashita K, Katagiri H, et al. Preoperative serum CA19-9 and dissected peripancreatic tissue margin as determiners of long-term survival in pancreatic cancer. Ann Surg Oncol 2009;16:1231-1240. [PubMed]
- Turrini O, Schmidt CM, Moreno J, et al. Very high serum CA 19-9 levels: a contraindication to pancreaticoduodenectomy? J Gastrointest Surg 2009;13:1791-1797. [PubMed]
- Wasan HS, Springett GM, Chodkiewicz C, et al. CA 19-9 as a biomarker in advanced pancreatic cancer patients randomised to gemcitabine plus axitinib or gemcitabine alone. Br J Cancer 2009;101:1162-1167. [PubMed]
- Katz MH, Varadhachary GR, Fleming JB, et al. Serum CA 19-9 as a marker of resectability and survival in patients with potentially resectable pancreatic cancer treated with neoadjuvant chemoradiation. Ann Surg Oncol 2010;17:1794-1801. [PubMed]
- Montgomery RC, Hoffman JP, Riley LB, Rogatko A, Ridge JA, Eisenberg BL. Prediction of recurrence and survival by post-resection CA 19-9 values in patients with adenocarcinoma of the pancreas. Ann Surg Oncol 1997;4:551-556. [PubMed]
- Hernandez JM, Cowgill SM, Al-Saadi S, et al. CA 19-9 velocity predicts disease-free survival and overall survival after pancreatectomy of curative intent. J Gastrointest Surg 2009;13:349-353. [PubMed]
- Nishida K, Kaneko T, Yoneda M, et al. Doubling time of serum CA 19-9 in the clinical course of patients with pancreatic cancer and its significant association with prognosis. J Surg Oncol 1999;71:140-146. [PubMed]
- Ishii H, Okada S, Sato T, et al. CA 19-9 in evaluating the response to chemotherapy in advanced pancreatic cancer. Hepatogastroenterology 1997;44:279-283. [PubMed]
- Gogas H, Lofts FJ, Evans TR, Daryanani S, Mansi JL. Are serial measurements of CA19-9 useful in predicting response to chemotherapy in patients with inoperable adenocarcinoma of the pancreas? Br J Cancer 1998;77:325-328. [PubMed]
- Halm U, Schumann T, Schiefke I, Witzigmann H, Mössner J, Keim V. Decrease of CA 19-9 during chemotherapy with gemcitabine predicts survival time in patients with advanced pancreatic cancer. Br J Cancer 2000;82:1013-1016. [PubMed]
- Stemmler J, Stieber P, Szymala AM, et al. Are serial CA 19-9 kinetics helpful in predicting survival in patients with advanced or metastatic pancreatic cancer treated with gemcitabine and cisplatin? Onkologie 2003;26:462-467. [PubMed]
- Ziske C, Schlie C, Gorschlüter M, et al. Prognostic value of CA 19-9 levels in patients with inoperable adenocarcinoma of the pancreas treated with gemcitabine. Br J Cancer 2003;89:1413-1417. [PubMed]
- Ko AH, Hwang J, Venook AP, Abbruzzese JL, Bergsland EK, Tempero MA. Serum CA19-9 response as a surrogate for clinical outcome in patients receiving fixed-dose rate gemcitabine for advanced pancreatic cancer. Br J Cancer 2005;93:195-199. [PubMed]
- Pohlank K, Hilbig A, Pelzer U, et al. Decrease of CA 19-9 in patients with advanced pancreatic cancer (APC) undergoing chemotherapy predicts survival time J Clin Oncol 2008;S26:15574. [abstract].
- Reni M, Cereda S, Balzano G, et al. Carbohydrate antigen 19-9 change during chemotherapy for advanced pancreatic adenocarcinoma. Cancer 2009;115:2630-2639. [PubMed]
- Hess V, Glimelius B, Grawe P, et al. CA 19-9 tumour-marker response to chemotherapy in patients with advanced pancreatic cancer enrolled in a randomised controlled trial. Lancet Oncol 2008;9:132-138. [PubMed]
- Fogelman DR, Pathak P, Qiao W, et al. Serum CA 19-9 level as a surrogate marker for prognosis in locally advanced pancreatic cancer (LAPC) J Clin Oncol 2008;S26:15514. [abstract].
- Haas M, Laubender RP, Stieber P, et al. Prognostic relevance of CA 19-9, CEA, CRP, and LDH kinetics in patients treated with palliative second-line therapy for advanced pancreatic cancer. Tumour Biol 2010;31:351-357. [PubMed]
- Takahashi H, Ohigashi H, Ishikawa O, et al. Serum CA19-9 alterations during preoperative gemcitabine-based chemoradiation therapy for resectable invasive ductal carcinoma of the pancreas as an indicator for therapeutic selection and survival. Ann Surg 2010;251:461-469. [PubMed]
- Willett CG, Daly WJ, Warshaw AL. CA 19-9 is an index of response to neoadjunctive chemoradiation therapy in pancreatic cancer. Am J Surg 1996;172:350-352. [PubMed]
- Boeck S, Haas M, Laubender RP, et al. Application of a time-varying covariate model to the analysis of CA 19-9 as serum biomarker in patients with advanced pancreatic cancer. Clin Cancer Res 2010;16:986-994. [PubMed]
- Kang CM, Kim JY, Choi GH, et al. The use of adjusted preoperative CA 19-9 to predict the recurrence of resectable pancreatic cancer. J Surg Res 2007;140:31-35. [PubMed]
- Decker GA, Batheja MJ, Collins JM, et al. Risk factors for pancreatic adenocarcinoma and prospects for screening. Gastroenterol Hepatol (N Y) 2010;6:246-254. [PubMed]
- Bedi MM, Gandhi MD, Jacob G, Lekha V, Venugopal A, Ramesh H. CA 19-9 to differentiate benign and malignant masses in chronic pancreatitis: is there any benefit? Indian J Gastroenterol 2009;28:24-27. [PubMed]
- Ulla Rocha JL, Alvarez Sanchez MV, Paz Esquete J, et al. Evaluation of the bilio-pancreatic region using endoscopic ultrasonography in patients referred with and without abdominal pain and CA 19-9 serum level elevation. JOP 2007;8:191-197. [PubMed]
- Paganuzzi M, Onetto M, Marroni P, et al. CA 19-9 and CA 50 in benign and malignant pancreatic and biliary diseases. Cancer 1988;61:2100-2108. [PubMed]
- Marcouizos G, Ignatiadou E, Papanikolaou GE, Ziogas D, Fatouros M. Highly elevated serum levels of CA 19-9 in choledocholithiasis: a case report. Cases J 2009;2:6662. [PubMed]
- Kim HR, Lee CH, Kim YW, Han SK, Shim YS, Yim JJ. Increased CA 19-9 level in patients without malignant disease. Clin Chem Lab Med 2009;47:750-754. [PubMed]
- Ventrucci M, Pozzato P, Cipolla A, Uomo G. Persistent elevation of serum CA 19-9 with no evidence of malignant disease. Dig Liver Dis 2009;41:357-363. [PubMed]
- Mery CM, Duarte-Rojo A, Paz-Pineda F, Gómez E, Robles-Díaz G. Does cholestasis change the clinical usefulness of CA 19-9 in pacreatobiliary cancer? Rev Invest Clin 2001;53:511-517. [PubMed]
- Ong SL, Sachdeva A, Garcea G, et al. Elevation of carbohydrate antigen 19.9 in benign hepatobiliary conditions and its correlation with serum bilirubin concentration. Dig Dis Sci 2008;53:3213-3217. [PubMed]
- Basso D, Fabris C, Plebani M, et al. Alterations in bilirubin metabolism during extra- and intrahepatic cholestasis. Clin Investig 1992;70:49-54. [PubMed]
- Marrelli D, Caruso S, Pedrazzani C, et al. CA19-9 serum levels in obstructive jaundice: clinical value in benign and malignant conditions. Am J Surg 2009;198:333-339. [PubMed]
- Ortiz-González J, Alvarez-Aguila NP, Medina-Castro JM. Adjusted carbohydrate antigen 19-9. Correlation with histological grade in pancreatic adenocarcinoma. Anticancer Res 2005;25:3625-3627. [PubMed]
- Koopmann J, Rosenzweig CN, Zhang Z, et al. Serum markers in patients with resectable pancreatic adenocarcinoma: macrophage inhibitory cytokine 1 versus CA19-9. Clin Cancer Res 2006;12:442-446. [PubMed]
- Eguchi H, Ishikawa O, Ohigashi H, et al. Serum REG4 level is a predictive biomarker for the response to preoperative chemoradiotherapy in patients with pancreatic cancer. Pancreas 2009;38:791-798. [PubMed]


